
R&D
We continue our efforts to discover new drugs based on our Corporate Philosophy “Dedicated to the Fight against Disease and Pain.”
The reason we strive in drug discovery is for patients battling with diseases that still have no treatment,
due to our strong desire to deliver innovative drugs as early as possible.
We continue striving to discover innovative new drugs to respond to the many unmet medical needs that exist in the world.
Ono’s Focus Fields
Tackling diseases that still lack effective treatments
Ono’s R&D aims at “creating unique and innovative new drugs,” focused on oncology, immunology & inflammation and neurology, which are our priority areas. We are deepening our understanding of human disease biology and leveraging digital technology to improve the speed and quality of drug discovery R&D while working to discover new drugs to respond to unmet medical needs.
Research

Corporate Officer / Executive Vice President, Discovery & Research
Message from the Executive Director of Discovery & Research
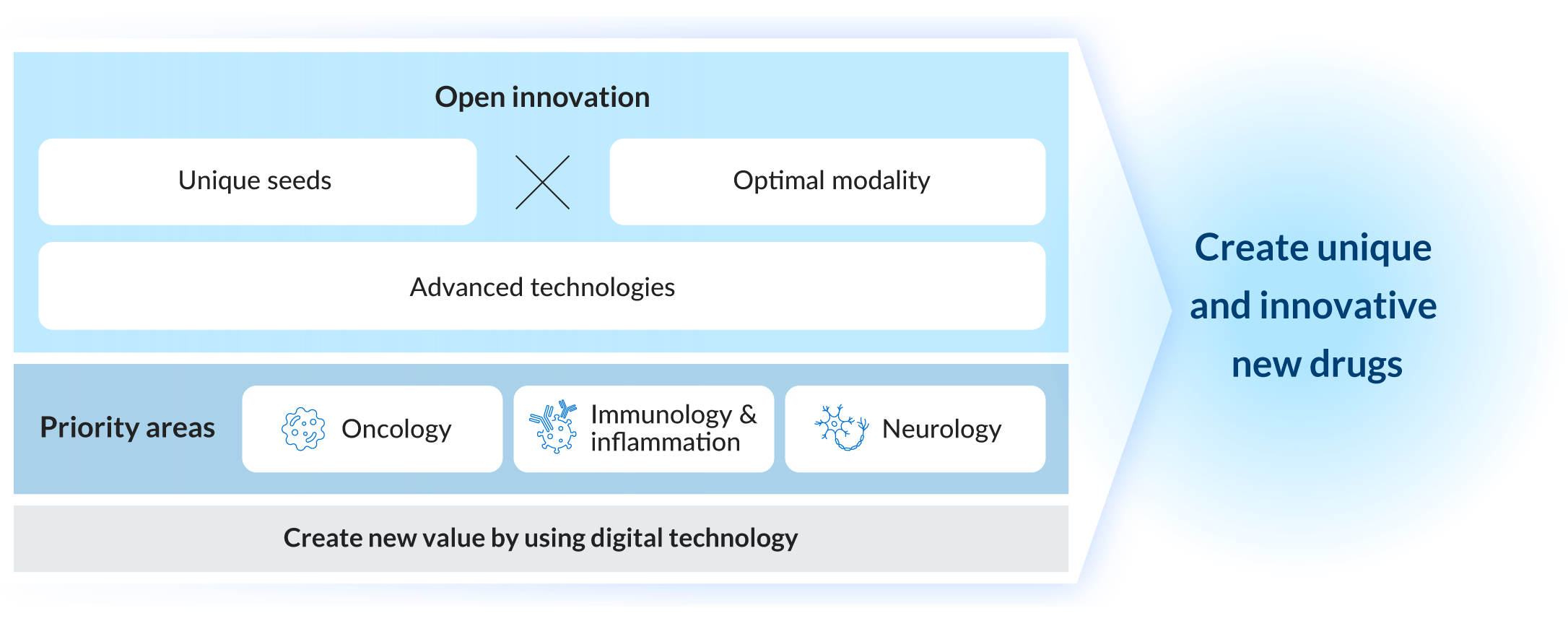
Taking on the Challenge of New Drug Discovery through Highly Proactive Open Innovation
Ono’s history in drug discovery is challenges. We are promoting highly proactive open innovation such as acquiring unique drug discovery seeds through research collaborations with academia, and creating innovative new drug candidates through drug discovery collaborations with bio-venture companies with cutting-edge technologies, aiming to create innovative new drugs that address unmet medical needs. Recently, we have been accelerating the search for new drug discovery seeds and shortening the time to creation of novel compounds by using Artificial Intelligence (AI) to analyze and utilize the vast amounts of information obtained from pioneering research, and with this are working to improve the success rate and speed of drug discovery.
Additionally, we are actively promoting opportunities for our researchers to visit our collaborative research institutions as visiting scientists, and to take up positions at our subsidiaries in the U.S. and Europe that explore opportunities for research and drug discovery collaborations, thereby raising the level of and engagement with each researcher. Going forward, we will further strengthen our systems to promote rapid, world-class drug discovery.
Drug discovery through co-creation and advanced technology
Highly Proactive Open Innovation
We are involved in drug discovery partnerships with research institutes, pharmaceutical companies, and bio-venture companies in Japan and overseas, chiefly in our priority areas.

Development

Corporate Officer / Executive Vice President, Clinical Development
Message from the Clinical Development Executive Director
Aiming to be a Global Specialty Pharma
—Pipeline Expansion, Maximizing Product Value, and Accelerating Global Development—
We are moving forward with clinical development in the key areas of oncology, immunology & inflammation and neurology, and focusing on pipeline reinforcement (strengthening), maximizing product value, and accelerating global development. To strengthen the pipeline, we are enhancing our ability to conduct trials in order to establish proof of concept (POC) at an early stage, and also incorporating various strategies to improve the accuracy of result
interpretation.
For products in the pipeline that have already been launched, we are maximizing product value by adding supplementary indications and developing new combination therapies to meet the diverse unmet needs that still exist.
Accelerating development speed—obtaining approval faster—is extremely important to maximize the value of new drug candidates. We maximized the development functions of Deciphera in the U.S. and Europe, which newly joined our Group last year, and will steadily proceed with international joint trials to obtain global approval as soon as possible.

BREAK THROUGH Story

Going and Beyond in Cancer Treatment

Delivering New Drugs to as Many Patients as Possible

Global Challenge to Acquire New Drug Candidate





