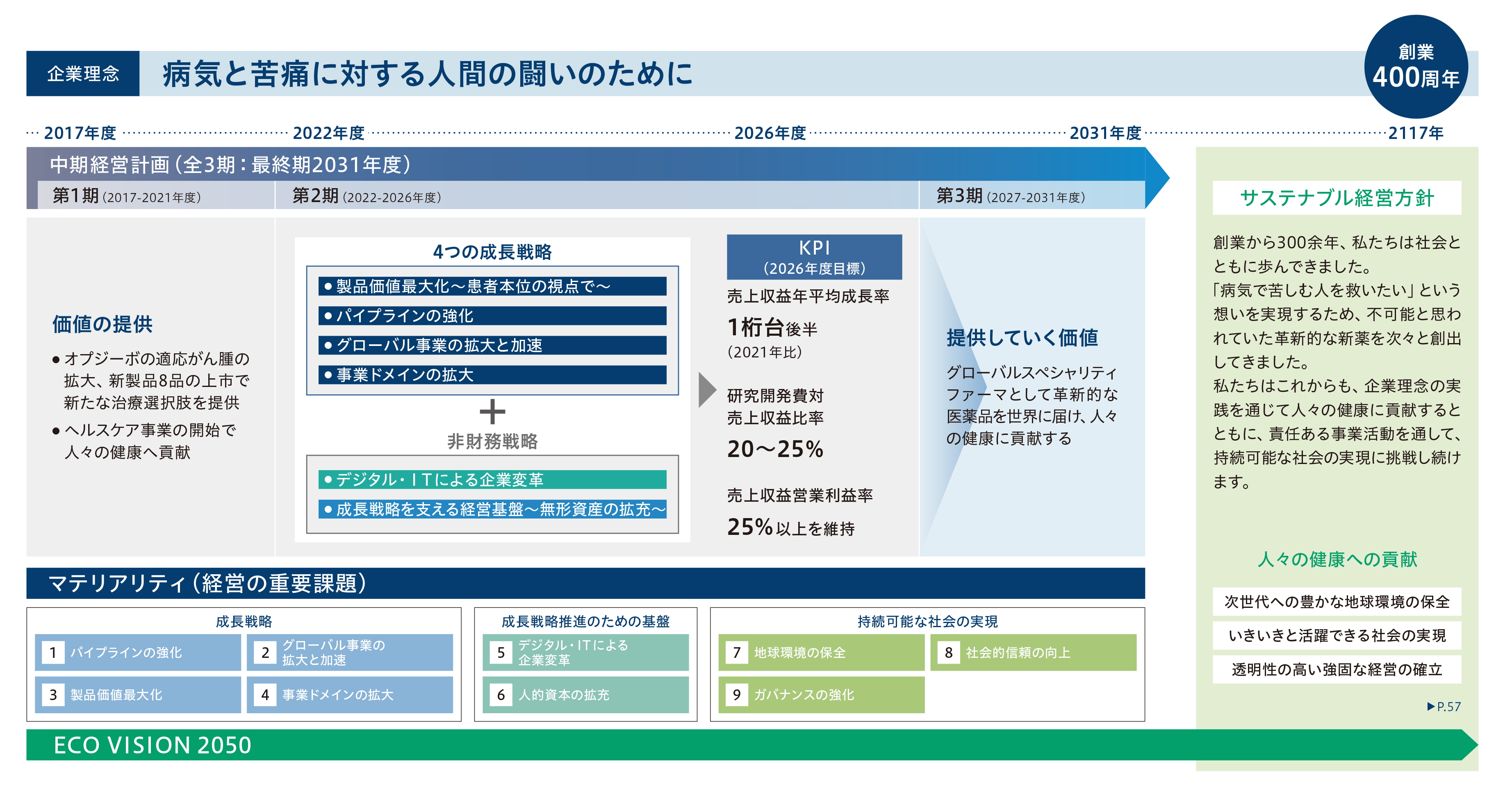
経営戦略
目指す未来へのロードマップ
創業300年を迎えた2017年度に、15年後の2031年度をゴールとする長期ビジョンを策定しました。目指すのは、革新的な医薬品を世界中に提供し続けるグローバル スペシャリティファーマです。「病気と苦痛に対する人間の闘いのために」という企業理念のもと、4つの成長戦略を定めて事業活動に取り組んでいます。さらに、これらの成長戦略を支える経営基盤である無形資産の拡充に努めています。わたしたちはこれからも、サステナブル経営方針のもと、次の100年を見据えて、持続可能な社会の実現に貢献し続けます。