グローバルスペシャリティファーマを目指して
執行役員/開発本部長
岡本 達也
グローバルスペシャリティファーマを目指して

執行役員/開発本部長
岡本 達也
-パイプラインの強化、製品価値最大化、グローバル開発の加速-
開発では重点領域に定めたがん、免疫・炎症、神経領域の疾患を対象とした臨床開発を推進しており、パイプラインの拡充(強化)、製品価値最大化およびグローバル開発の加速に取り組んでいます。パイプラインの強化においてはPOCの早期確立のために試験の実行力を強化するとともに、結果解釈の精度を高めるためのさまざまな工夫を取り入れています。すでに上市を果たしたパイプラインについては、未だ満たされない多様なアンメットニーズに応えるために効能・効果の追加や新規併用療法の開発に取り組むことをもって、製品価値の最大化につなげています。新薬候補の価値最大化にとって開発スピードの加速化、すなわち早期承認取得は非常に重要です。昨年、新たにグループに加わったデサイフェラ社の米欧における開発機能を最大限活用し、グローバルで一日でも早く承認を取得するための国際共同試験を着実に進めてまいります。
臨床開発
パイプラインの強化
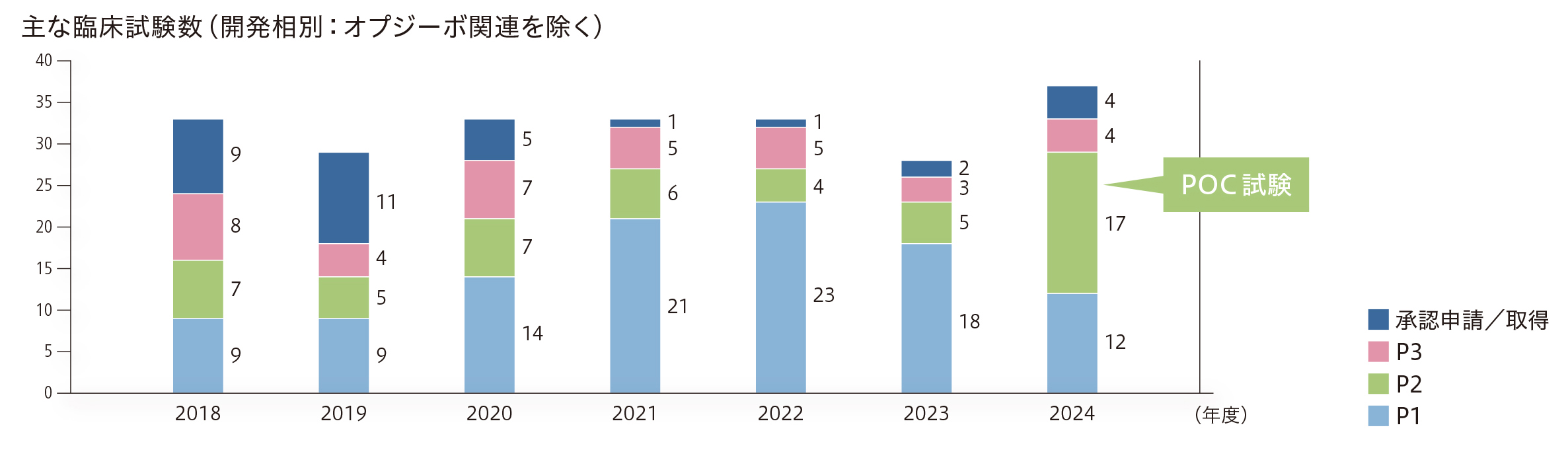
臨床開発においては、パイプラインの質的強化、すなわちPOCが確立された多くのプロジェクトを有し、たくさんの検証的位置づけの臨床試験が実施されている状態の実現を目指しています。自社の創薬研究で生み出された化合物やライセンス活動で獲得した化合物を、病気で苦しんでいる世界中の患者さんに1日も早くお届けするには、パイプラインの質的な強化が必要です。そのためには、POCを早期に確立することが重要であり、同時期に複数の疾患を対象とした臨床試験を並行して実施できる体制を構築して運用することで、臨床ステージに移行してからPOCの確立に至るまでの期間短縮に取り組んでいます。また、できるだけコンパクトな臨床試験で精度高くPOC確立を判断するための方策として、トランスレーショナルリサーチに基づくバイオマーカーの活用やリアルワールドデータ、自社で取得した臨床データの活用などにも積極的に取り組んでいます。
製品価値最大化
化合物の製品価値を高めるために、既存製品の効能追加にも取り組んでいます。対象疾患そのものを拡大することに加え、より早い治療段階から使用できるようにすることや、より治療効果を向上させることができる併用療法の確立など、アンメットニーズを満たすための開発を続けています。さらに、より利便性の高い投与方法の検討により患者さんの負担を軽減する策を講じています。
成長戦略の実現に向けて
グローバル開発の加速
これまで海外においては、自販体制を確立している韓国と台湾を除いて、自社創製の化合物の開発および販売をパートナー企業にライセンスアウトしてきました。しかし、これからは、世界最大のマーケットを持つ米国および欧州においても、当社が創製・開発した化合物を自らの手で患者さんにお届けするようにしたいと考えています。この目的を達成するために、これまで、米国および欧州における臨床試験の遂行はもちろんのこと、承認申請から承認取得に至るまでを含む臨床開発に係るすべての工程を担える体制の強化・整備を進めてきましたが、昨年、欧米における承認申請・取得の経験を持つデサイフェラ社が当社グループに加わったことから、欧米における臨床開発機能をデサイフェラ社に集約し、さらなるグローバル開発の加速を目指します。