積極的なライセンス活動の推進
自社での創薬研究によるパイプラインの拡充に加え、国内外の製薬企業やバイオベンチャー企業などとの革新的な医薬品の創製を目的とした創薬提携活動や、開発中の新薬候補化合物の導入をめざしたライセンス活動も積極的に進めています。導入に関しては、既存製品や開発パイプラインも考慮して事業戦略性・効率性が高いと判断された化合物、あるいは医療ニーズの高い疾患に対する魅力のある化合物に注目しています。
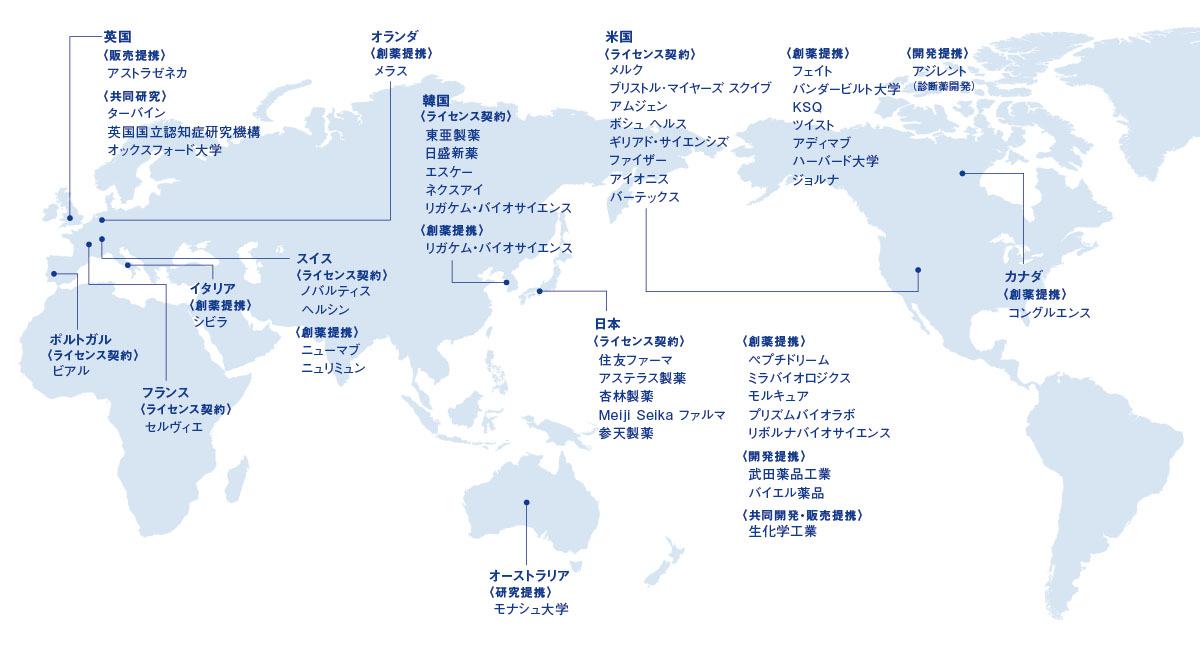
国内外の主な提携先 (2026年5月8日現在)
当社のこれまでの提携活動をご紹介します。国内外問わず、積極的な提携活動を推進しています。
提携企業
当社の主な提携先をご紹介します。
なお、企業ロゴをクリックすると各社との提携内容をご覧いただけます。
導入・導出・共同販促
導入・導出・共同販促
ブリストル・マイヤーズ スクイブ社 (米国)

2005年5月にメダレックス社(ブリストル・マイヤーズ スクイブ社(BMS社)が買収)とヒト型抗ヒトPD-1(programmed cell death-1)モノクローナル抗体に関する研究提携契約を締結し、その研究から生まれた「ニボルマブ(ONO-4538/BMS-936558/MDX-1106)」を創製しました。2011年9月に、ニボルマブの全世界での開発を加速させるために、BMS社の開発・販売テリトリーを日本・韓国・台湾を除く全世界に拡大する提携契約を締結しました。また、2014年7月には、日本、韓国および台湾において戦略的提携契約を締結し、複数の免疫療法薬について単剤および併用療法として共同で開発・商業化に取り組んでいます。当社は、2014年9月に日本で悪性黒色腫の治療薬として「オプジーボ®点滴静注」を発売後、さまざまながん腫での承認を取得しています。
また、ニボルマブに関する契約締結と同時に、当社は、BMS社の関節リウマチ治療剤である「オレンシア®(アバタセプト)」を日本においてBMS社の日本子会社であるブリストル・マイヤーズ スクイブ株式会社と共同で開発・販売する契約を締結し、2013年8月に「オレンシア®皮下注」の製品名で新発売しました。
ノバルティス社 (スイス)

2005年12月にノバルティス社とアルツハイマー型認知症治療剤リバスチグミン(ONO-2540/ENA713D)の日本における開発・販売に関する契約を締結しました。ノバルティス社の子会社であるノバルティス ファーマ株式会社と共同開発後、2011年7月に「軽度及び中等度のアルツハイマー型認知症における認知症症状の進行抑制」の効能又は効果で、当社は「リバスタッチ®パッチ」、ノバルティスファーマ株式会社は「イクセロン®パッチ」の製品名で新発売しました。本剤は、アルツハイマー型認知症治療剤としては初めて経皮吸収型貼付剤にしたものです。
ヘルシングループ (スイス)

2006年10月に米国のサファイア社(現在、スイスのヘルシングループの一企業であるHelsinn Healthcare SAが継承)と同社が開発中のがん悪液質治療剤アナモレリン(ONO-7643)を日本、韓国および台湾で独占的に開発・販売するライセンス契約を締結しました。アナモレリンは経口グレリン様作用薬で、がん悪液質の患者さんにおける体重および筋肉量の増加並びに食欲の増加効果を示しています。当社は、2021年1月に日本で「悪性腫瘍(非小細胞肺癌、胃癌、膵癌、大腸癌)におけるがん悪液質」の効能又は効果で製造販売承認を取得し、同年4月に「エドルミズ®錠」の製品名で新発売しました。
アムジェン社 (米国)

2010年9月にオニキス社(現アムジェン社の子会社)と、二つのプロテアソーム阻害剤「カルフィルゾミブ(ONO-7057)およびオプロゾミブ(ONO-7058)」について、全がん腫を対象に日本において独占的に開発・販売する契約を締結しました。カルフィルゾミブについては、2012年7月に米国で再発又は難治性の多発性骨髄腫の治療薬としてカイプロリス®の製品名で上市されました。日本では、2016年7月に同適応症で製造販売承認を取得し、同年8月に「カイプロリス®点滴静注用」の製品名で新発売しました。2019年11月に、既承認のデキサメタゾンとの併用療法による週 2 回投与に加え、同剤との併用療法による週1回投与の用法及び用量の追加承認を取得し、服薬利便性が向上しています。
2011年9月にカイ社(後にアムジェン社が買収)と、維持透析下の二次性副甲状腺機能亢進症を対象に開発中のカルシウム受容体作動薬「エテルカルセチド塩酸塩(ONO-5163)」について、日本で独占的に開発・販売する契約を締結しました。本剤は、副甲状腺にあるカルシウム受容体に作用して、副甲状腺ホルモンの過剰な分泌を抑制させることで、血中のカルシウム値およびリン値を低下させ、動脈硬化などの心血管系障害の発症リスクを低下させます。2016年11月に欧州で、2017年2月に米国で「慢性腎臓病に伴う血液透析患者における二次性副甲状腺機能亢進症」の適応で承認を取得しています。日本では2016年12月に「血液透析下の二次性副甲状腺機能亢進症」の適応で製造販売承認を取得し、2017年2月に「パーサビブ®静注透析用」の製品名で新発売しました。
セルヴィエ社 (フランス)

2011年9月にセルヴィエ社と、「イバブラジン塩酸塩(ONO-1162)」について、日本で独占的に開発・販売する契約を締結しました。イバブラジンは、心臓のペースメーカー機能を担うイオンチャネルの一つであるHCNチャネルを選択的に阻害し、心臓の収縮機能や血圧に影響を及ぼさずに心拍数を低下させる薬剤です。本剤は、2006年1月にセルヴィエ社により安定狭心症の治療薬として海外で発売され、2012年2月に欧州、2015年4月には米国で慢性心不全の治療薬として承認されました。日本では、当社が2019年9月に「洞調律かつ投与開始時の安静時心拍数が75回/分以上の慢性心不全(ただし、β遮断薬を含む慢性心不全の標準的な治療を受けている患者に限る。)」の効能又は効果で製造販売承認を取得し、同年11月に「コララン®錠」の製品名で新発売しました。
ビアル社 (ポルトガル)

2013年4月に、ビアル社が「パーキンソン病における症状の日内変動(wearing-off現象)」の治療薬として海外で開発中の長時間作用型COMT(カテコール-O-メチルトランスフェラーゼ)阻害剤「オピカポン(ONO-2370)」について、日本で独占的に開発・販売するライセンス契約を締結しました。本剤は、これまでの臨床試験において1日1回の服用により持続的なCOMT阻害活性が示されており、服薬利便性の向上が期待されます。欧州では、2016年6月に同剤は「レボドパ/ドパ脱炭酸酵素阻害剤(DCI)併用療法で症状が安定しないwearing-off現象が認められるパーキンソン病の患者さんにおける補助療法」として承認され、「オンジェンティス®」の製品名で販売されています。日本では、当社が2020年6月に「レボドパ含有製剤との併用によるパーキンソン病における症状の日内変動(wearing-off現象)の改善」の効能又は効果で製造販売承認を取得し、同年8月に「オンジェンティス®錠」の製品名で新発売しました。
アストラゼネカ社 (英国)

2013年12月に、アストラゼネカ社と、ナトリウム・グルコース共輸送体2(SGLT-2)阻害剤であるダパグリフロジンについて、日本におけるコ・プロモーション契約を締結しました。当社は本剤「フォシーガ®(ダパグリフロジン)錠」の日本での流通・販売を担当し、両社で共同販促を行っています。フォシーガ®は、腎尿細管でのグルコース再吸収を制御するSGLT-2に対する選択的かつ可逆的な阻害剤であり、血液中の過剰なグルコースを尿と共に体外へ排出させ、血糖を低下させる薬剤です。本剤は、インスリンを介さずに空腹時血糖および食後の高血糖を改善する2型糖尿病治療薬として承認された世界で最初のSGLT-2 阻害剤です。2014年5月に、1日1回経口投与の2型糖尿病治療薬として「フォシーガ®錠」の製品名で新発売しました。2019年3月に1型糖尿病、2020年11月に標準治療を受けている慢性心不全、2021年8月には末期腎不全又は透析施行中の患者を除く慢性腎臓病に対する効能又は効果の追加承認を取得しました。
ギリアド・サイエンシズ社 (米国)

2019年7月に米国フォーティーセブン社(2020年4月にギリアド社の完全子会社となる)と、同社が様々ながん腫を対象に開発中の抗CD47抗体であるmagrolimab(ONO-7913)について、日本、韓国、台湾およびASEAN諸国で独占的に開発および商業化するライセンス契約を締結しました。Magrolimabは、マクロファージ上のSIRPαとがん細胞上のCD47の結合を阻害する抗CD47モノクローナル抗体で、がん細胞がマクロファージからの貪食作用を回避する「don’t eat me」シグナルを無効化します。当社は日本でTP53変異陽性急性骨髄性白血病を対象とした第Ⅲ相臨床試験を骨髄異形成症候群、膵がん、結腸・直腸がんを対象とした第Ⅰ相臨床試験を実施しています。なお、ギリアド社は、海外で骨髄異形成症候群や急性骨髄性白血病などを対象に開発を進めています。
ファイザー社 (米国)

2017年5月にアレイバイオファーマ社(現ファイザーの子会社)と、BRAF阻害剤「エンコラフェニブ(ONO-7702)」とMEK阻害剤「ビニメチニブ(ONO-7703)」について、日本および韓国で独占的に開発・商業化するライセンス契約を締結しました。ビラフトビ®(エンコラフェニブ)およびメクトビ®(ビニメチニブ)は、悪性黒色腫をはじめ種々のがんに関連するMAPKシグナル伝達経路(RAS-RAF-MEK-ERK)のセリン・トレオニンキナーゼファミリーの異なるキナーゼ、BRAFおよびMEK1/MEK2をそれぞれ標的として選択的に阻害し、がん細胞の増殖を抑制します。当社は、2019年1月に日本で「BRAF遺伝子変異を有する根治切除不能な悪性黒色腫」の効能又は効果で両製剤の製造販売承認を取得し、同年2月に発売しました。2020年11月には、両製剤と抗ヒトEGFRモノクローナル抗体であるセツキシマブとの3剤併用療法およびビラフトビ®とセツキシマブとの2剤併用療法における「がん化学療法後に増悪したBRAF遺伝子変異を有する治癒切除不能な進行・再発の結腸・直腸癌」の効能又は効果の追加承認を取得しました。2023年5月、日本でビラフトビとメクトビの併用によるBRAF遺伝子変異を有する根治切除不能な甲状腺がんに対する効能又は効果の追加に係る製造販売承認事項一部変更承認申請を行いました。
生化学工業株式会社 (日本)

2017年8月に生化学工業株式会社と、同社が開発中の変形性関節症治療剤SI-613の日本における共同開発および販売提携に関する契約を締結しました。生化学工業は、糖質科学を専門分野とする研究開発型の製薬企業です。1947 年の創業以来、糖質科学の可能性に着目し、運動器疾患や眼科疾患領域において独創的で有用な医薬品・医療機器を創製し続けています。ONO-5704/SI-613(ジクロフェナクエタルヒアルロン酸ナトリウム)は、生化学工業独自の薬剤結合技術を用いてヒアルロン酸とジクロフェナク(抗炎症薬)を化学結合した薬剤です。2021年3月に生化学工業が「変形性関節症(膝関節、股関節)」の効能又は効果で製造販売承認を取得し、同年5月に当社が「ジョイクル®関節注30mg」の製品名で新発売しました。
また、2025年8月には、生化学工業株式会社が開発を進めている変形性関節症治療剤「Gel-One」の日本における共同開発および販売提携に関する契約を締結しました。
「Gel-One」は、日本において変形性関節症(膝関節)を対象とした第Ⅲ相臨床試験、変形性関節症(股関節)を対象とした第Ⅲ相臨床試験、および安全性評価を主目的とする両関節を対象とした長期投与試験を実施中です。
コーナーストーン社 (米国)

2019年6月にラファエル社(2022年5月にコーナーストーン社に名称変更)と、同社が開発中のがん代謝阻害剤であるdevimistat(ONO-7912)およびその関連化合物について、日本、韓国、台湾およびASEAN諸国での独占的な開発および商業化を目的とするライセンス契約を締結しました。コーナーストーン社は、がん代謝阻害剤の分野を牽引している企業で、治療困難ながんに有効な新規の治療薬の開発に取り組んでいます。devimistatは、細胞のミトコンドリア分画に存在し、細胞の増殖および生存に必要であるトリカルボン酸(TCA)サイクルを標的としてがん細胞を選択的に阻害し、がん細胞において様々な化学療法剤に対する感受性を大幅に高めます。この相乗効果により、低用量の化学療法剤とdevimistatを併用することで、化学療法剤の治療で一般的に認められる副作用を軽減しつつ有効性を高めることができると期待されます。
エスケー社 (韓国)

2020年10月に、SK Biopharmaceuticals Co., Ltd. (SKBP社)と同社の抗てんかん薬であるcenobamate(ONO-2017)について、日本において独占的に開発および商業化するライセンス契約を締結しました。SKBP社は、中枢神経系(CNS)疾患の治療薬の研究、開発および商業化に注力するグローバル製薬企業で、てんかんを含むCNS疾患の治療薬として8つの開発化合物で構成されるパイプラインを有しています。Cenobamateは、SKBP社とその米国子会社であるSK Life Science社によって創製・開発されました。Cenobamateが治療的効果をもたらす詳細な作用機序は不明ですが、cenobamateは電位依存性ナトリウム電流を阻害することにより、反復的な神経発火を減らすと考えられています。また、γアミノ酪酸 A型(GABAA)イオンチャネルの正の調節作用があります。Cenobamateは、2020年5月より米国で成人患者におけるてんかん部分発作の治療薬として、XCOPRI®の商品名で販売されています。また、2021年3月に欧州においても販売承認されました。日本では、てんかん強直間代発作、てんかん部分発作を対象とした第Ⅲ相臨床試験を実施しています。Cenobamateは、日本のてんかん患者さんの新たな治療選択肢になると期待されます。
コーディア社 (日本)

2020年12月に、Chordia Therapeutics株式会社(コーディア社)と、同社の粘膜関連リンパ組織リンパ腫転座1 (MALT1)阻害剤であるCTX-177およびその関連化合物について、全世界での独占的な開発、製造および商業化を目的とするライセンス契約を締結しました。コーディア社は、2017年11月にがん領域に特化した研究開発型バイオベンチャー企業として設立され、新しい作用機作を有する抗がん薬の研究と開発を行い、革新的な新薬を生み出すことを目指しています。MALT1の活性化は、リンパ球系の血液細胞のがん化に重要であることが報告されており、CTX-177 は MALT1 の活性を選択的に阻害することにより、リンパ球系の血液腫瘍に対する抗腫瘍効果が期待されます。
ライボン社 (米国)

2021年2月にライボン社と、同社が開発中のPARP7(ポリADPリボースポリメラーゼ7)阻害剤であるAtamparib(ONO-7119)について、日本、韓国、台湾およびASEAN諸国での独占的な開発および商業化を目的とするライセンス契約を締結しました。ライボン社は、細胞がストレスを受けた際に活性化する酵素を標的にしたファーストインクラスの薬剤を開発するバイオテクノロジー企業です。治療選択肢が限られた患者に有効な治療法を届けるべく、独自の創薬プラットフォームであるBEACON+を活用して新たな治療薬の開発に取り組んでいます。Atamparibは、がん細胞の生存にとって重要な役割を果たす分子であるPARP7を阻害することで、腫瘍増殖を直接抑えることに加え、がん細胞に対する免疫応答を高めるという2つのメカニズムを有することから、新たながん治療薬になることが期待されます。現在、当社は日本で固形がんを対象とした第Ⅰ相臨床試験を実施しています。
NEX-I社(韓国)

2024年3月、NEX-I社と、「NXI-101」に関するライセンス契約を締結しました。NXI-101は、NEX-I社独自の標的探索プラットフォーム(ONCOKINE®プラットフォーム)により同定されたがん免疫療法抵抗性因子ONCOKINE-1に対するファーストインクラスの抗体医薬であり、がん免疫療法抵抗性のがんを含むさまざまながん腫に対する有効性が期待されます。当社はNXI-101およびそのバックアップ抗体を全世界で独占的に開発・商業化する権利を取得します。
アイオニス社(米国)

2025年3月、米国Ionis Pharmaceuticals Inc.(Ionis社)と、同社が真性多血症の治療薬として開発中の「sapablursen」について、全世界を対象に独占的な開発および商業化するライセンス契約を締結しました。Ionis社はRNA標的医薬品のパイオニアであり、30年以上にわたり、重篤な疾患を抱える人々により良い未来をもたらす医薬品の創製に取り組んできました。Ionis社は、6つの承認された治療薬と神経、循環器、患者ニーズの高い特定領域における先進的な開発化合物パイプラインを有しています。「sapablursen」は、Ionis社によって創製・開発されたTMPRSS6を標的とした核酸医薬(アンチセンス阻害剤)です。TMPRSS6の産生を抑制し、鉄恒常性の主要な調節因子であるヘプシジンの産生を増加させることにより、真性多血症(PV)をはじめとして様々な血液疾患の治療薬となる可能性があります。Ionis社は、PVを対象とした第Ⅱ相試験の完了まで引き続き開発を担当します。当社は、その後の全世界を対象としたsapablursenの開発および商業化活動を担当します。
バーテックス社(米国)

2025年6月、米国Vertex Pharmaceuticals Incorporated(Vertex社)と、IgA腎症、原発性膜性腎症を含む複数のB細胞介在性疾患の治療薬として開発中の「Povetacicept」について、日本、韓国を対象に独占的な開発および商業化するライセンス契約を締結しました。Vertex社は、重篤な疾患に対する革新的な医薬品を創出するための科学的イノベーションに投資するグローバルバイオテクノロジー企業です。「Povetacicept」は、遺伝子組換え融合タンパク質であり、IgA腎症治療においてベストインクラスのポテンシャルを有するBAFF(B cell activating factor)およびAPRIL(a proliferation inducing ligand)に対するデュアル拮抗薬であり、IgA腎症および原発性膜性腎症患者を対象とした臨床試験において、ベストインクラスの有効性を示す可能性が示されています。また、Povetaciceptは他の自己免疫性腎疾患および自己免疫性血球減少症を含む複数の重篤なB細胞介在性疾患の治療薬としても開発中です。
メルク社 (米国)
2004年11月にメルク社より新規の経口2型糖尿病治療剤シタグリプチン(ONO-5435/MK-0431)と癌化学療法に伴う制吐剤 アプレピタント(ONO-7436/MK-0869)を導入しました。
シタグリプチンはDPP-4(ジペプチジルペプチターゼ-4)に対する選択的阻害剤で、血糖値を下げる生体内の仕組み(インクレチンシステム)を活性化することにより血糖値をコントロールする糖尿病治療薬で、日本では、当社と米メルク社の子会社であるMSD株式会社が共同開発し、2009年12月に当社は「グラクティブ錠」、MSD株式会社は「ジャヌビア錠」の製品名で発売しました。なお、欧米では米メルク社が「JANUVIA」の製品名で販売しています。
アプレピタントは、選択的ニューロキニン1受容体拮抗剤で、抗悪性腫瘍剤の投与に伴う急性及び遅発性嘔吐に有効で、日本では、当社が単独で開発し、2009年12月に「イメンドカプセル」の商品名で新発売しました。なお、欧米ではメルク社が「EMEND」の製品名で販売しています。また、2011年12月に当社は、イメンドの有効成分であるアプレピタントをプロドラッグ化した注射剤である「プロイメンド点滴静注用」を新発売し、2016年3月には生後6ヵ月以上12歳未満の小児患者さんへの使用も可能となりました。経口剤である「イメンドカプセル」の服薬が困難である患者さんがおられることや、点滴静注で投与される抗がん剤も多いことから、抗悪性腫瘍剤投与に伴う悪心・嘔吐の治療に新たな選択肢となっています。
ボシュ ヘルス社 (米国)
2013年10月にバリアント社(2018年7月にボシュ ヘルス社に名称変更)と、「デムサー®(メチロシン)」について、日本で独占的に開発・商業化する契約を締結しました。デムサーは、カテコールアミンの産生に関わるチロシン水酸化酵素を阻害することで、褐色細胞腫から過剰に分泌されるカテコールアミンを減少させ、カテコールアミン過剰分泌による高血圧、頻脈、不整脈などの症状を軽減します。日本では、当社が2019年1月に「褐色細胞腫のカテコールアミン分泌過剰状態の改善」の効能又は効果で製造販売承認を取得し、同年2月に「デムサー®カプセル」の製品名で発売しました。
