医薬品の安心・安全を保証するために
当社は、患者さんの立場に立ち、医薬品のライフサイクルを通じてグローバルな観点で信頼性保証活動を行っており、医薬品の品質が確保されていることを常に確認するとともに、医療従事者や患者さんの声を一層の品質改善につなげています。さらに、患者さんや医療従事者からの報告、文献や調査などから収集した、医薬品の品質および有効性、安全性(副作用)に関わる情報を、最先端の科学に基づいて分析・評価し、常に最新の情報を医療の現場に提供しています。
また当社は医薬品の品質、安全性確保等を目的として以下の業界団体に所属するとともに、業界が定める医薬品の適正使用に関するコードやガイドラインを遵守し、信頼性保証活動を推進しています。
- 日本製薬工業協会
- 関西医薬品協会
- 医療用医薬品製造販売業公正取引協議会
- くすりの適正使用協議会
- 日本QA研究会
品質保証に関する取り組み
当社は、品質保証にかかる全社方針として「クオリティ グローバル ポリシー」を制定し、国内外の開発から市販後に至る医薬品ライフサイクルを通じた品質、安全性および有効性を担保するためのコミットメントを明文化して、患者さんの安全とウェルビーイングに貢献するよう取り組んでいます。品質・安全性リスクは重大なリスクとして認識し、全社のERMに統合して管理しています。また、医薬品製造業者、医薬品製造販売業者としての法的要件にとどまらず、グローバルに医薬品品質システムを確立するために、ICHガイドラインに則って品質マニュアルを策定し、システムの継続的な改善を図っています。加えて、品質管理システムを内部監査する部門として、GxP実施部門から独立した品質保証統括部にグローバルクオリティオーディット部を設置し、第三者として内部監査を実施しています。そして、品質が高度に保証された医薬品を安定的に供給することにより、社会に貢献します。このような取り組みの結果、2024年度は当社製品回収がありませんでした。
また、社員一人ひとりが品質や安全性への意識を高めるよう、製造販売三役体制による適切な管理体制のもと、全従業員を対象とし、医薬品に関わる法令遵守教育ならびに薬害教育を定期的に実施しています。
医薬品の適正使用のための取り組み
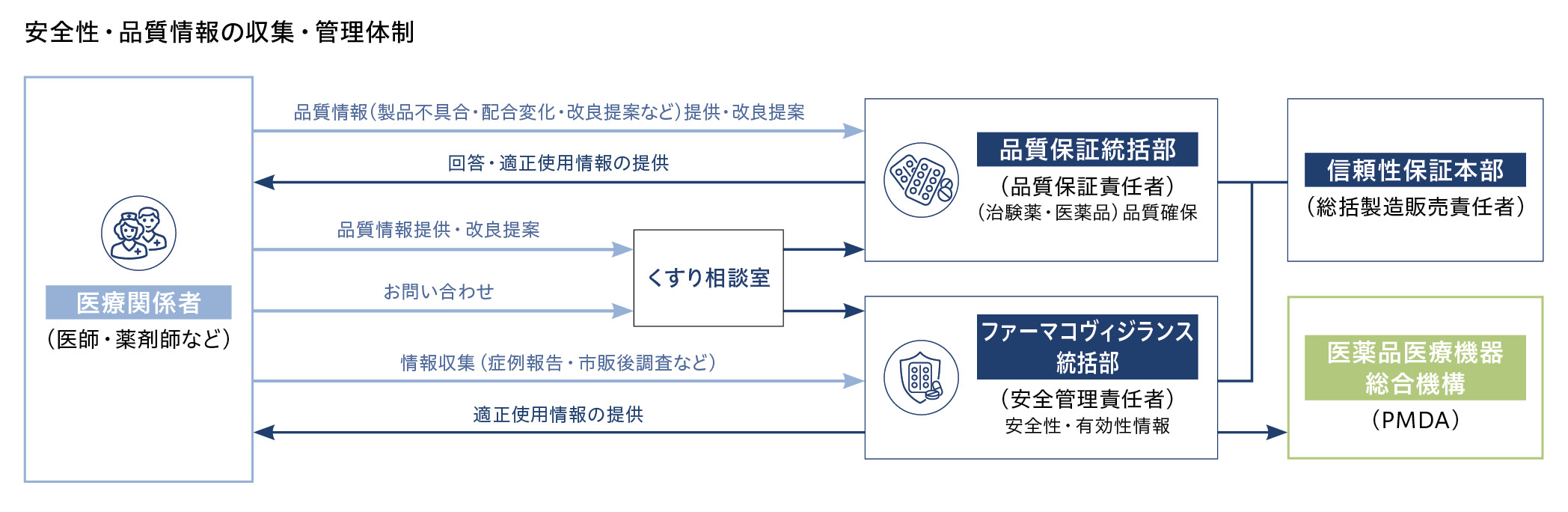
当社は、医薬品ごとのリスク管理計画策定と、安全性(副作用)情報の収集および管理を行っています。
収集した情報の内容を評価し、必要に応じて添付文書の「使用上の注意」の改訂や医薬品の適正使用に関するお知らせの提供などの安全性対策を実施しています。
抗悪性腫瘍剤の上市後は国内外からの安全性情報が劇的に増加しており、それらを社外の医学専門家の意見を踏まえて評価したうえで、各種情報提供資材や学会・医学雑誌を通じて発信するなど、適正使用を推進しています。
安全管理活動においても、グローバルでの標準手順書やデータベースを整備し、海外を含むグループ全体の体制を構築しています。
製品回収体制の整備
製品の有効性、品質、安全性に問題がある場合は、その情報を速やかに医療従事者に提供し、当該製品を回収する体制を整えています。また、製品の回収を想定した訓練を関連する部署が合同で定期的に実施することによって、不測の事態にも迅速に対応できる体制であることを確認しています。
